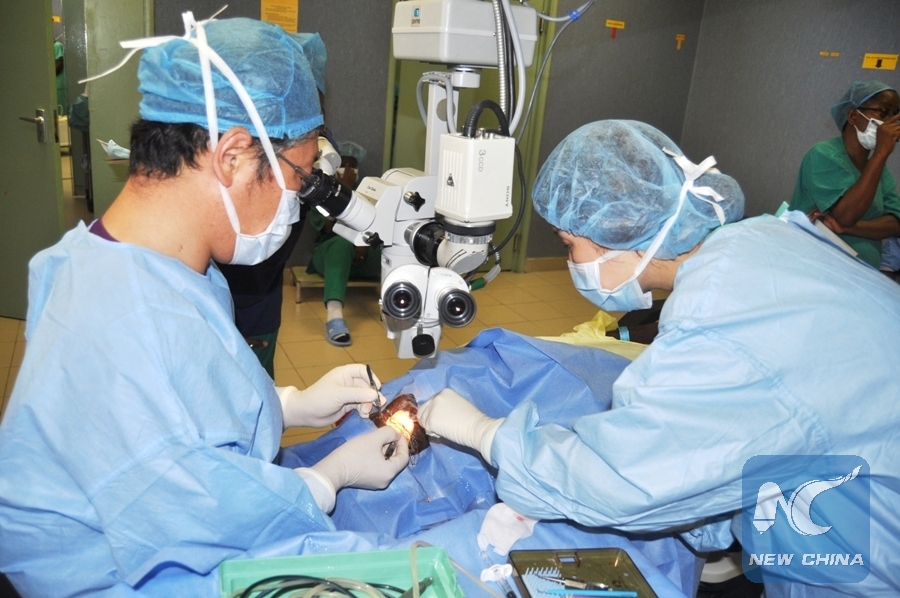
International Blindness Relief Organization Vision Care President Dr. Dong Hae Kim (L) performs an eye cataract surgery during a three-day free eye surgical camp activity at Muhimbili National Hospital in Dar es Salaam, Tanzania, Aug. 9, 2016. The activity is held in partnership with Muhimbili University of Health and Allied Sciences (MUHAS). More than 25 patients underwent eyes surgery within two days. (Xinhua/John Badi)
WASHINGTON, Jan. 3 (Xinhua) -- Luxturna, the first gene therapy approved in the United States for a rare, inherited form of blindness, will cost 850,000 U.S. dollars per patient, its maker announced on Wednesday.
"The Wholesale Acquisition Cost of one-time LUXTURNA is 425,000 dollars per eye," Monique da Silva, a spokeswoman for Sparks Therapeutics, told Xinhua in an email.
"The price reflects the stakeholder considerations we have learned about these past months, including access concerns of patients, families, health care providers and treatment centers."
While the price came in lower than the one-million-dollar estimate by U.S. analysts, it's still among the most expensive treatments in the world.
Spark Therapeutics said it will offer rebates to health insurers if the treatment does not work.
The company said it has reached agreement in principle with Boston-based insurer Harvard Pilgrim to offer the outcomes-based rebate arrangement and is also in active discussions with other commercial insurers regarding this offering.
Spark Therapeutics also said it's addressing budgetary concerns by working towards multiple solutions to allow installment payments over multiple years.
An international research team led by Chinese scientists has found a new gene that can cause congenital blindness. (Xinhua file photo by Lin Hong)
Luxturna was approved last month by U.S. Food and Drug Administration (FDA) to treat children and adult patients with confirmed biallelic RPE65 mutation-associated retinal dystrophy that leads to vision loss and may cause complete blindness in certain patients.
It works by delivering a normal copy of the RPE65 gene directly to retinal cells, leading these cells to produce the normal protein that converts light to an electrical signal in the retina to restore patient's vision loss.
Prior to this approval, the FDA greenlighted two other gene therapies that re-engineer people's own immune cells to treat blood cancers.
Both came with hefty price tags. Yescarta, a drug that treats certain types of large B-cell lymphoma, has a price tag of 373,000 dollars. Kymriah, a leukemia therapy, is similarly expensive at 475,000 dollars.

